Challenge

A customer developing a new SPF50 suncream spray needed to validate the stability of their formulations before committing to any reformulation decisions and subsequent scale-up. Traditional stability testing meant months of delay, with no early indication of where formulations were failing. They needed a way to benchmark their stability and identify any weaknesses compared to established market products quickly.
Solution
Instead of relying on long-term stability studies, we benchmarked their formulations against a leading market competitor using a suite of our complementary rheological and interfacial techniques to build a robust stability profile for each sample:
- Zeta Potential – Electrostatic repulsion between particles
- Zero shear viscosity – Viscosity at close to at-rest conditions
- Yield Stress & Structure – The stress required to initiate flow
This combined approach accelerates the development cycle by providing actionable data in a matter of days, rather than months.
Business Impact
At the Centre for Industrial Rheology, we routinely carry out stability testing for a range of different products with our suite of complementary techniques. Whether you are benchmarking new candidates against existing products or troubleshooting a formulation, our testing removes the guesswork and gives your development team the confidence to make faster, better-informed decisions. We provide:
- Rapid Turnaround – a comprehensive stability characterisation study delivered in 1-2 weeks
- Early Risk Identification – pinpoint formulation weaknesses before committing to scale-up or long-term storage studies
- Formulation Benchmarking – rank your formulations directly against market products to understand where you stand
- Reformulation confidence – the data we deliver informs where development effort may need to be focused
Results
The testing revealed clear and actionable differences between formulations.
- Sample A (Strongest Performer) – This formulation performed well against the market benchmark. It demonstrated a slightly higher zeta potential and yield stress, with a slightly lower zero-shear viscosity.
- Sample B (Moderate Risk) – Although this sample maintained a high zeta potential, both its yield stress and zero-shear viscosity were much lower. This indicates that it may be less stable compared to both the market benchmark and sample A.
- Sample C (High Risk) – This was highlighted as the biggest “danger” formulation, performing significantly worse across all three metrics. Its zeta potential dropped to just -18.61 mV, its yield stress was notably lower, and its zero-shear viscosity was an order of magnitude below the other samples.
Importantly, the strongest performing samples still exhibited the shear-thinning behaviour required for effective spraying, confirming that stability and sprayability are not mutually exclusive with the right formulation decisions.
The full analytical data supporting these findings are presented in the Analytical Findings section.
Extending The Study
As these are sprayable suncare formulations, future work can look at capturing and benchmarking the primary contributors to spraying performance. Specifically looking at:
- Dynamic Surface Tension at Short Surface Ages
- Shear Thinning Behaviour
- Thixotropic Behaviour
Analytical Findings
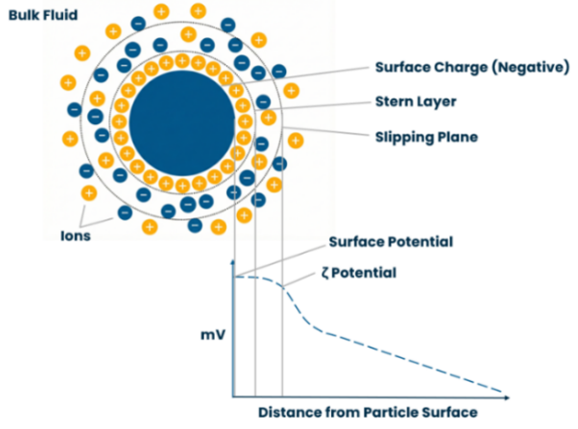
Every particle in a suspension or droplet in an emulsion develops a surface charge. The environment surrounding the particle is visualised in the concept of the electrical double layer:
- Stern Layer – A layer of ions oppositely charged to the particle, bound tightly to the surface.
- Diffuse Layer – A secondary layer of ions that are loosely associated, forming a cloud around the particle.
Within the diffuse layer, a theoretical boundary exists, known as the Slipping Plane. Zeta potential is the electrical potential measured specifically at this slipping plane. Utilising our high-concentration cell attachment, we can measure samples with little to no dilution. Where dilution is required, we aim to dilute with a dispersant of matching pH and conductivity.
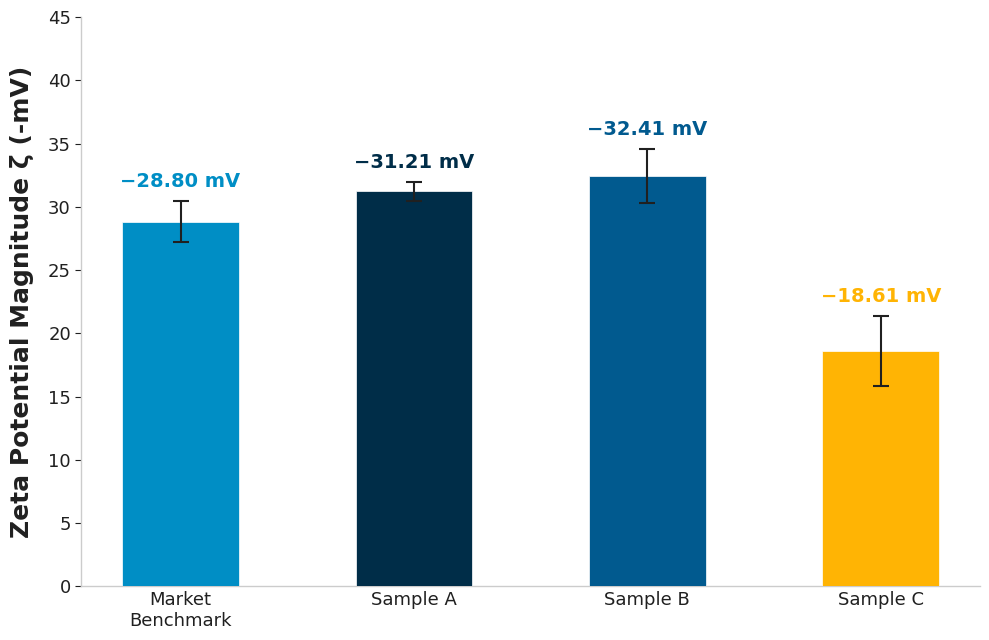
In colloidal science, a zeta potential magnitude below -30 mV often indicates a system that is prone to instability. This is due to the repulsive barrier being weak, and over time, Brownian motions will lead to collisions between particles, resulting in flocculation or coalescence. Over time, these events can manifest as visible phase separation, compromising the product’s long-term stability.
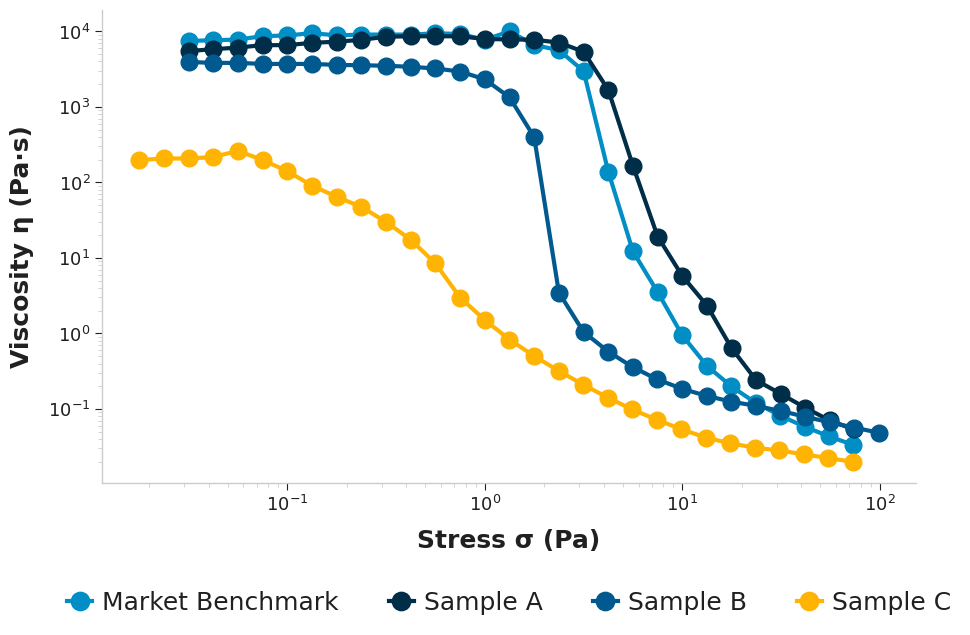
Zero-Shear Viscosity Measurements
The zero-shear viscosity reflects the viscosity of a product when it is effectively at rest. It is arguably one of the most practical indicators of stability, yet it is frequently overlooked as viscometers, and even some rheometers, lack the torque sensitivity to perform such tests. Our research rheometers in the lab offer extraordinary sensitivity, detecting low stresses that extend far beyond the range of typical viscometers. They are capable of detecting rotational speeds as slow as 1 revolution every 3 months. Despite this, we obtain rapid results in as little as 30 minutes. A high zero-shear viscosity in the external phase can be desirable, as it helps to immobilise particles and droplets.
The zeta potential results highlight a clear differentiation in the electrostatic repulsion between particles of these formulations. Sample C exhibits a notably lower magnitude, with a mean zeta potential of -18.61 mV. In contrast, samples A and B maintain much higher values around the -30 mV threshold.
The market benchmark exhibited the highest zero-shear viscosity, whereas Sample C showed a significantly lower zero-shear viscosity than all other samples. Despite these stark disparities at rest, the high-shear viscosity values converged significantly; notably the market benchmark (0.0335 Pa.s) and sample C (0.02 Pa.s) displayed comparable behaviour under high-shear conditions. This underscores that a high zero-shear viscosity, essential for stability at rest, does not inherently compromise a product’s sprayability or spreadability. The market benchmark, in particular, demonstrates a superior shear-thinning profile.
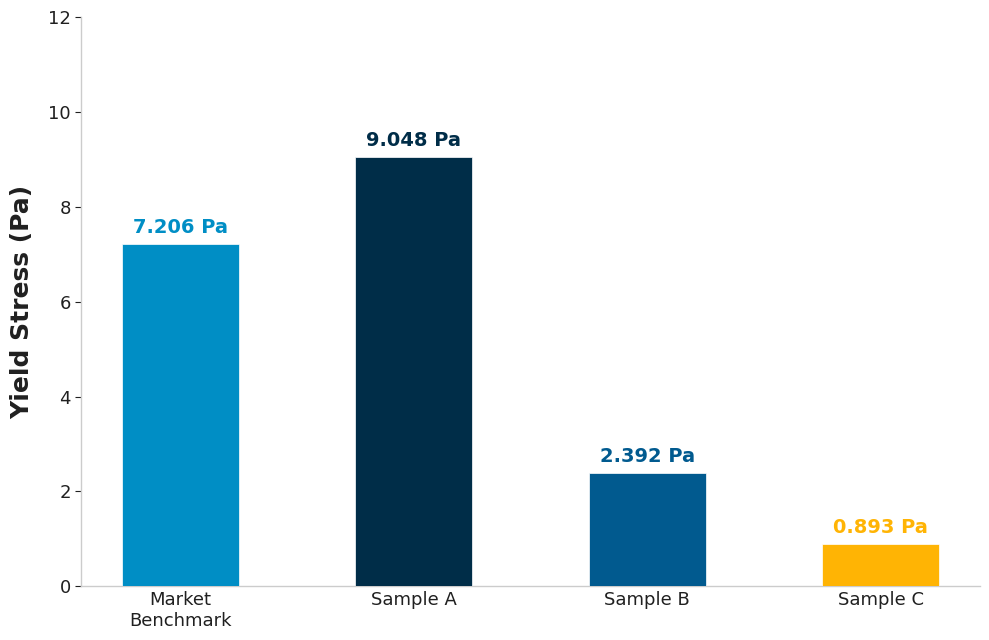
Yield Stress & Structure Measurements
To get further insights into stability, we go beyond viscosity to look at viscoelasticity and the concept of yield stress. Yield stress is the minimum amount of stress required to make a material flow. Think of it as the “strength” of the internal network when the product is sitting still. A sufficient yield stress acts as a physical net that immobilises particles or droplets. If the yield stress is strong enough to counteract the gravitational force exerted by the dispersed particles, sedimentation and creaming are effectively inhibited, aiding long-term shelf stability.
To measure this, we employ oscillatory techniques, where we gently “wobble” the sample to allow us to probe the delicate internal structure present without destroying it. A key metric in this analysis is the phase angle. This metric quantifies the material’s state under oscillatory stress, with a phase angle of 0° indicating a purely elastic solid, while 90° indicates a purely viscous liquid.
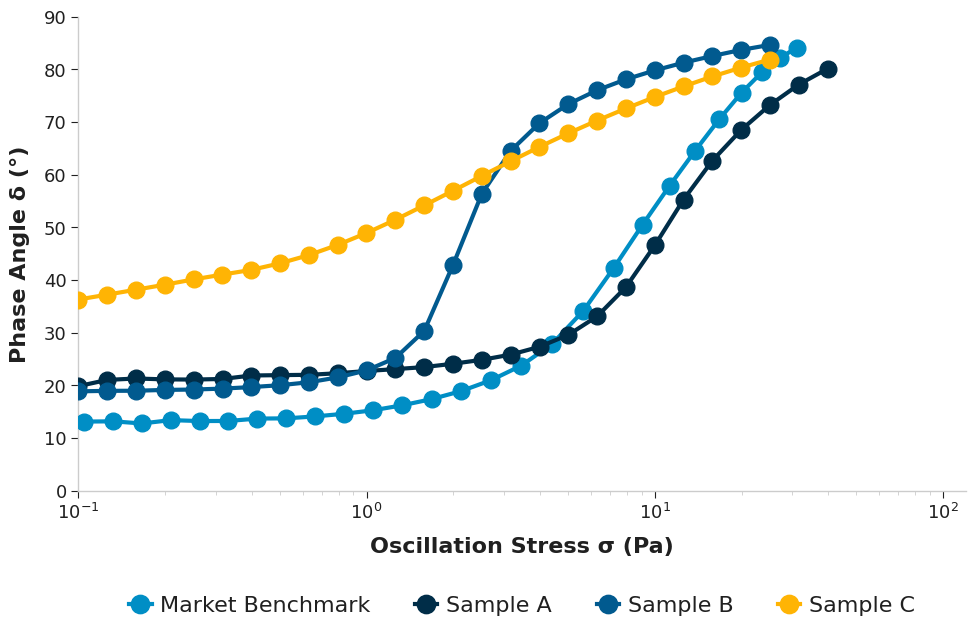
The market benchmark displayed the lowest phase angle at low stresses, indicating a more elastic structure at rest compared to the others. Sample A exhibited the highest yield stress, which suggests better shelf-life stability. Sample C’s lower yield stress can be seen as an advantage through an easier actuation force for spraying; however, as seen with the zero-shear viscosity and zeta potential data, it is compromising stability at rest in the bottle.
Summary
At the Centre for Industrial Rheology, we can help to remove the guesswork from formulation development by utilising our wide suite of synergistic techniques. Whether you are looking to benchmark your product or troubleshoot an issue, we provide high-precision testing to give you the data you need to formulate with confidence.

Related Articles;
Optimising Rheology and Dynamic Surface Tension for Sprayable Suncare Formulations
Predicting Formulation Stability: Advanced Insights for Suspensions and Emulsions
Wasif Altaf serves as an Applications Specialist at the Centre for Industrial Rheology, leveraging a chemical engineering background (BEng) to bridge theory and practice. His work focuses on advanced rheological characterisation.
