The delivery of drugs to the eyes is fraught with difficulty due to the body’s biological defence mechanisms. Barriers include blinking and wash-out by the tears, nasolacrimal drainage, non-productive losses and impermeability of the cornea. Rheology is an established technique for profiling the mucoadhesive properties of ophthalmic drops and is a valuable tool for determining if a solution is likely to have a longer residence time in the eye, which can lead to enhanced ophthalmic uptake and increased bioavailability.
Contact us to arrange a lab tour and discuss rheological techniques with our experts
 Ophthalmic drug administration: challenges and solutions
Ophthalmic drug administration: challenges and solutions
There are three main ways to administer drugs ophthalmically:
- Topically;
- Systemically;
- Intraocularly.
If the drug molecule is able to bypass the body’s immediate defence mechanisms, navigating the structure of the cornea is the next challenge. The cornea itself is comprised of five layers, which either have hydrophilic or hydrophobic (alternatively thought of as lipophilic) properties. Designing a drug that can cross both hydrophilic and hydrophobic barriers is especially difficult.
There are several ways that drug formulations can be adapted to improve the bioavailability of the material:
- Adding a solubility enhancer; this allows the concentration of a drug within a formulation to be increased, therefore smaller droplets can be used within each application.
- The formulation can be designed to resist clearance, for example by use of mucoadhesive technologies.
- Add a drug-penetration enhancer to enhance transportation across the cornea.
Mucoadhesion

Mucoadhesion is defined as ‘the state in which two biological materials adhere to each other for extended periods of time with the help of interfacial forces’.
Designing drugs that are mucoadhesive can afford significant advantage. For example, if the drug is to be administered in an area that is likely to secrete mucus, engineering mucoadhesive characteristics into a formulation allows the drug to interact with the mucus secreted, therefore the drug stays within the target area. This can increase residence time, leading to an enhanced likelihood of drug penetration.
Within the ophthalmic market there are a number of mucoadhesive materials that can be used. On the open market the most commonly used mucoadhesive materials contained within OTC ophthalmic formulations are carbomer and hyaluronic acid. This study identified differences in mucoadhesion between formulations using rheology. Four samples were tested:
- Sample A, containing 2% w/v hyaluronic acid
- Sample B, containing 0.15% w/v hyaluronic acid
- Sample C, containing glycerol
- Sample D, containing 0.32% w/v hypromellose
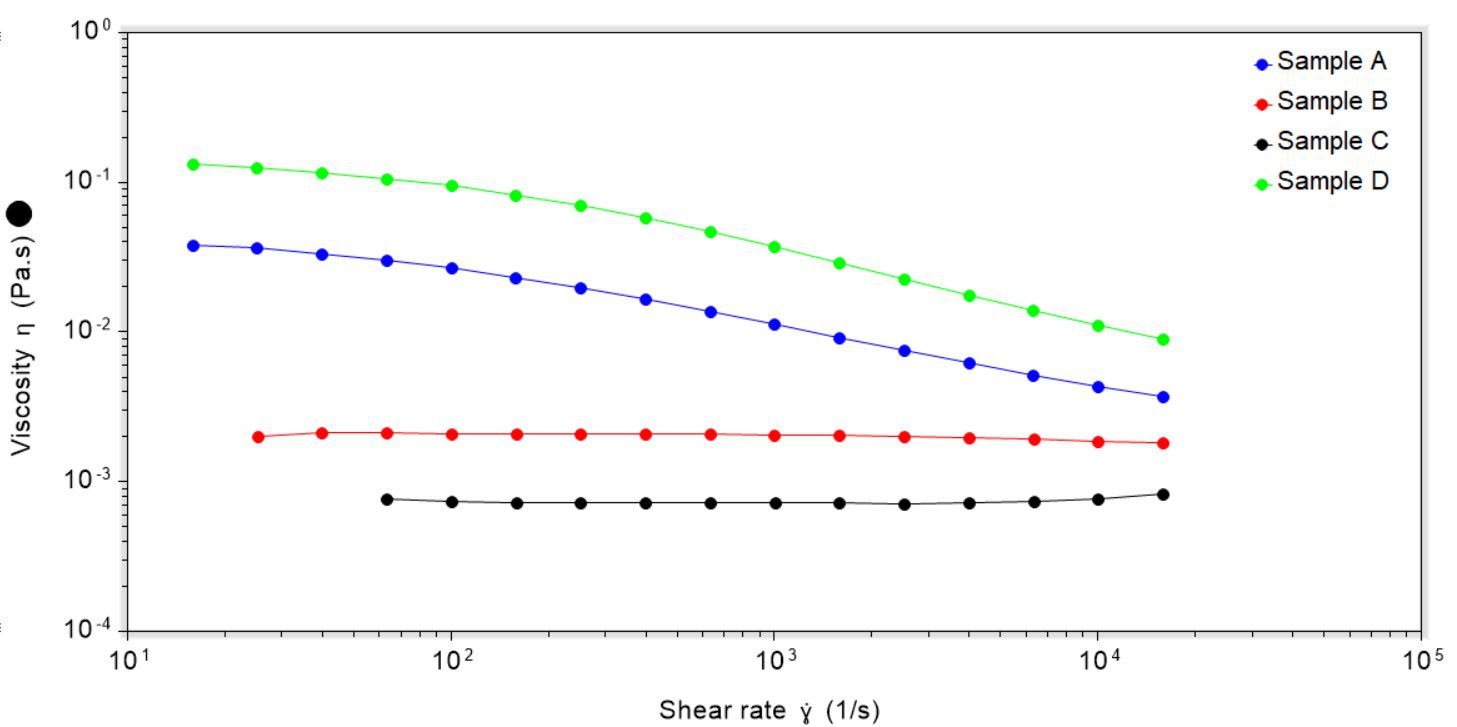
In terms of rheological testing, a controlled rate viscosity profile (CRVP) was collected for each sample to determine the impact of shear rate upon viscosity. A CRVP experiment allows for characterisation of a materials under a tightly controlled set of shear conditions, which is particularly important for topical eye formulations at both low and high shear rates. At low shear rates, a high viscosity will help maintain persistence of the film in the eye. However, during high shear events, such as blinking, a high viscosity will lead to a ‘draggy’ feel, or like a foreign body is trapped in the eye. This means that as the shear rate is increased the material should ideally decrease in viscosity. This can be described as ‘non-Newtonian, shear thinning’ behaviour.
Considering the CRVP data for the four samples tested, it can quickly be seen that samples A and D were shear-thinning, i.e. as the shear rate increased the viscosity decreased, Figure 1. Samples B and C were both Newtonian in behaviour, and a change in shear rate did not significantly affect the viscosity.
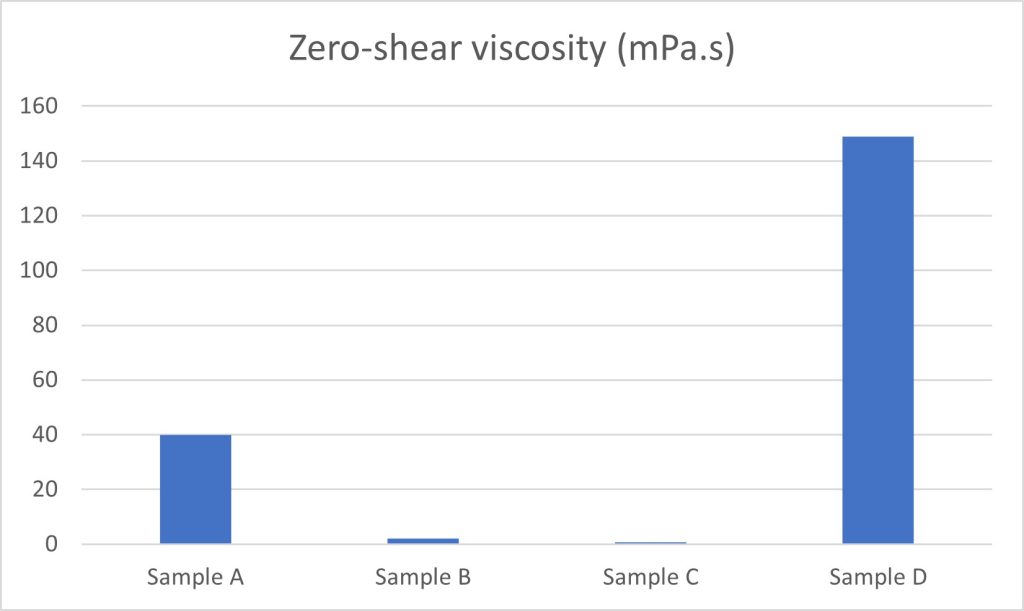
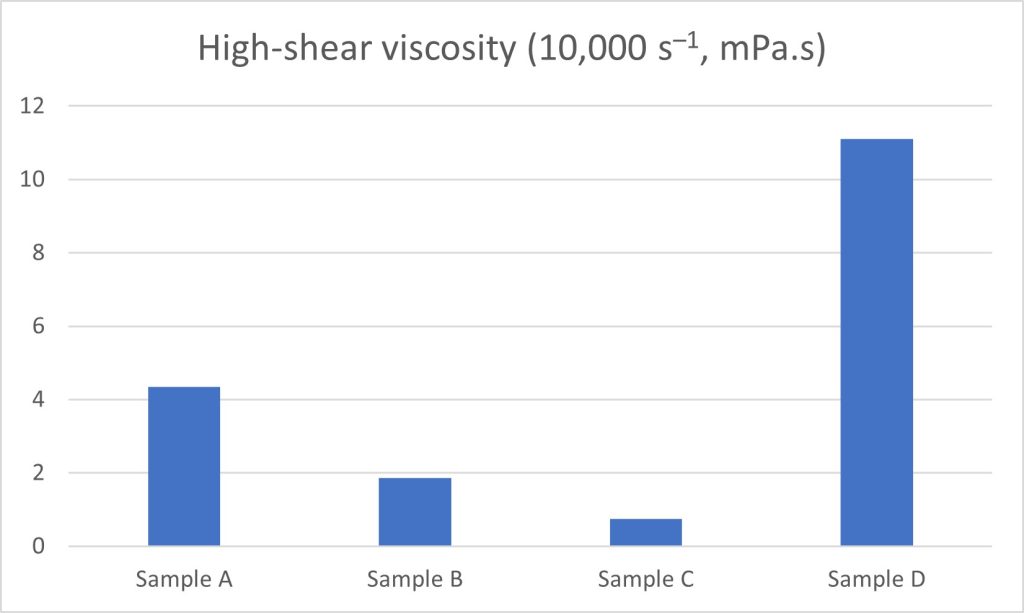
Across all shear rates the most viscous sample was D and the least viscous sample was C. The implication of this is that at low shear rates (i.e. when the eye is open) sample D will have a high residence time with low drainage, but at higher shear rates sample D may feel ‘draggy’. In comparison, sample C may drain from the eye quickly at low shear rates, but at higher shear rates (i.e. when blinking) may feel more natural than sample D.
When plotted as a bar chart, clear differences in eye drop behaviour can be noted at both zero-shear (i.e. when at rest, determined using the Cross model) and high shear (10,000 s–1) rates, Figures 2 and 3:
Rheology to Determine mucoadhesive characteristics
Interaction of eye drops with porcine gastric mucin (a commonly used surrogate for fluids such as tears, vaginal secretions and saliva) can also be determined using CRVP profiling. In this case, three sets of data are required:
- Mucin with deionised (DI) water
- Product with DI water
- Product with mucin
These three sets of data allow for investigation into whether the change in viscosity of a product upon addition of mucin is down to dilution effects (that are identified by mixing the product with DI water and, separately, mixing the mucin with DI water and measuring the viscosity) or a true change in viscosity due to positive or negative interactions between the mucin and sample; any ‘additional’ viscosity observed in the mucin and sample mix, over and above the sum of the viscosities of the separate solutions, suggests a synergistic interaction between the product and the mucin. This based upon work by Hassan et al and Madsen et al.
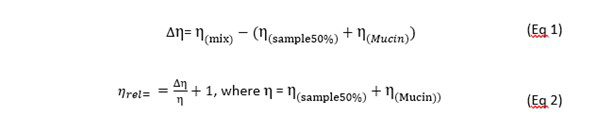
A change in the relative viscosity of over 1 shows a positive interaction between the mucin and sample, and therefore a high degree of interaction between mucin and the sample can be inferred. In this case, the high degree of interaction could indicate mucoadhesion, which could lead to a longer residence time for the drug and the longer that a drug is in contact with a tissue, the more likely it is to penetrate.
This data shows that sample A shows a strong positive interaction with mucin, whereas sample B shows less of an interaction and samples C and D show no interaction, Figure 4.
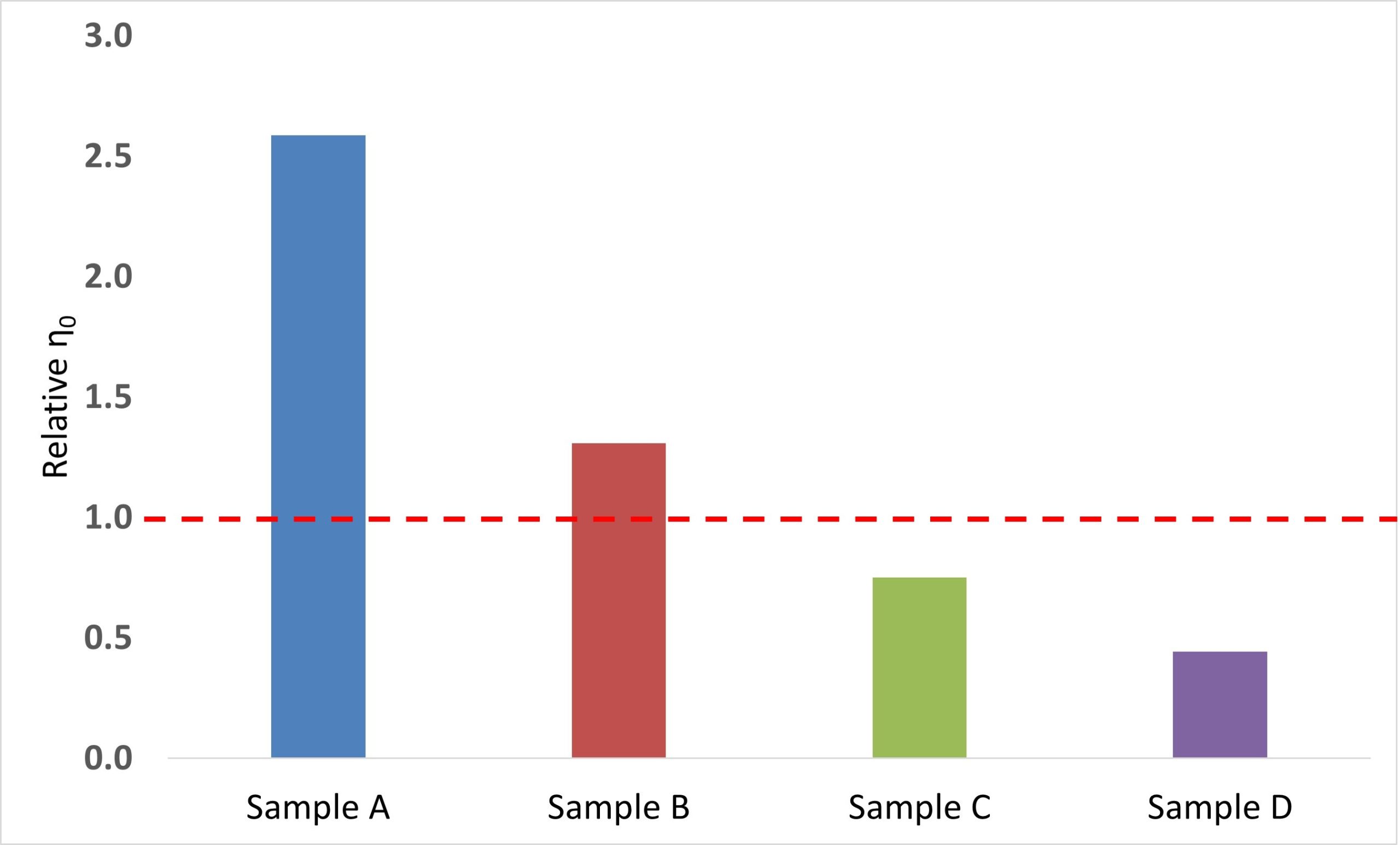
Within this sample-set it could be concluded that hyaluronic acid leads to a positive interaction with mucin as both samples A and B contain hyaluronic acid (2% w/v and 0.15% w/v, respectively), whereas samples C and D do not. In addition, it can be postulated at an increased amount of hyaluronic acid in the sample leads to a larger mucoadhesive response.
In summary, rheology, in particular viscosity profiling, is a valuable technique for probing how mucin interacts with biological fluids such as tears, vaginal secretions and saliva. This work can provide a solid basis for mucoadhesion work, streamlining the R+D process and providing quantitative data.
For further information about this study, and to find out more about the testing services that we offer, please contact us now!
