 Thermal conductivity measurements can tell you how well a material is able to conduct or transfer thermal energy, and specific heat capacity describes the quantity of thermal energy required to increase the temperature of a material. Both metrics are vital for predicting the performance of insulators, phase change materials such as oils, waxes and plastics, heat exchangers, evaporators, refrigerators and freezers.
Thermal conductivity measurements can tell you how well a material is able to conduct or transfer thermal energy, and specific heat capacity describes the quantity of thermal energy required to increase the temperature of a material. Both metrics are vital for predicting the performance of insulators, phase change materials such as oils, waxes and plastics, heat exchangers, evaporators, refrigerators and freezers.
Thermal conductivity and specific heat capacity measurements are routine in multiple industries including aerospace, automotive, construction, electronics, textiles, and additive manufacturing. Having a good understanding of how thermal energy conduction and how this affects a material is key to being able to predict the behaviour of a system and design and optimise controls. Our lab can measure the thermal conductivity of fluids, pastes, powders, insulators, and ceramics from -20 up to 200°C.
Contact us for a chat or prices
Thermal Conductivity Measurement Methods – Transient Plane Source (TPS) and Modified Transient Plane Source
We use the Transient Plane Source (TPS) approach for measuring thermal conductivity and its associated properties. A pulse of heat is applied to the sample and its temperature change is monitored with time. In the standard transient plane source method, two sheets of sample sit above and below the heater/sensor. For “through-plane” conductivity measurements, heat can be drawn through the sample sheets by adding highly-conducting steel “pucks” on the outside of the sample sheets. If “in-plane” measurements are required, thermally insulating blocks replace the steel pucks. The Modified Transient Plane Source method (MTPS), the sample sits atop the sensor. A cell is available to hold powder or liquid samples.

The process of heat transfer seems intuitive at first glance, but can be easy to overlook if you’re not careful – if you’ve ever had a burnt barbecue, then you’ve already seen how low thermal conductivity and low heat capacity can be a challenge to deal with, when working with high temperatures. In industry, overlooking thermal responses of your product can be a costly mistake, leading to inefficiencies, discrepancies in quality, or at worst blockages or damage to your process line.
Thermal Conductivity
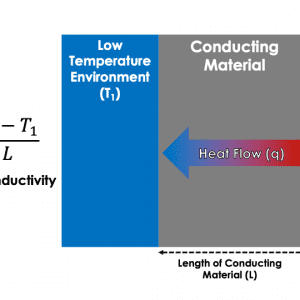
Thermal conductivity describes the rate of heat flow across a temperature gradient within a material; in other words, the ease at which thermal energy flows from the hot end to the cold end of the material. The SI unit for thermal conductivity is Watts per metre Kelvin (W/m·K).
A material with a high thermal conductivity can move vast amounts of heat quickly across a large distance, whereas a material with a low thermal conductivity can act as an insulating barrier to heat transfer. Thermal conductivity is vital for thermal interface materials which must be capable of facilitating heat transfer between two surfaces. It can be incredibly difficult to get two surfaces to have perfect contact with each other (and thus difficult to achieve perfect heat transfer efficiency) so thermal interface materials are used to ‘fill in the gaps’ and provide a route for heat transfer.
Thermal Diffusivity
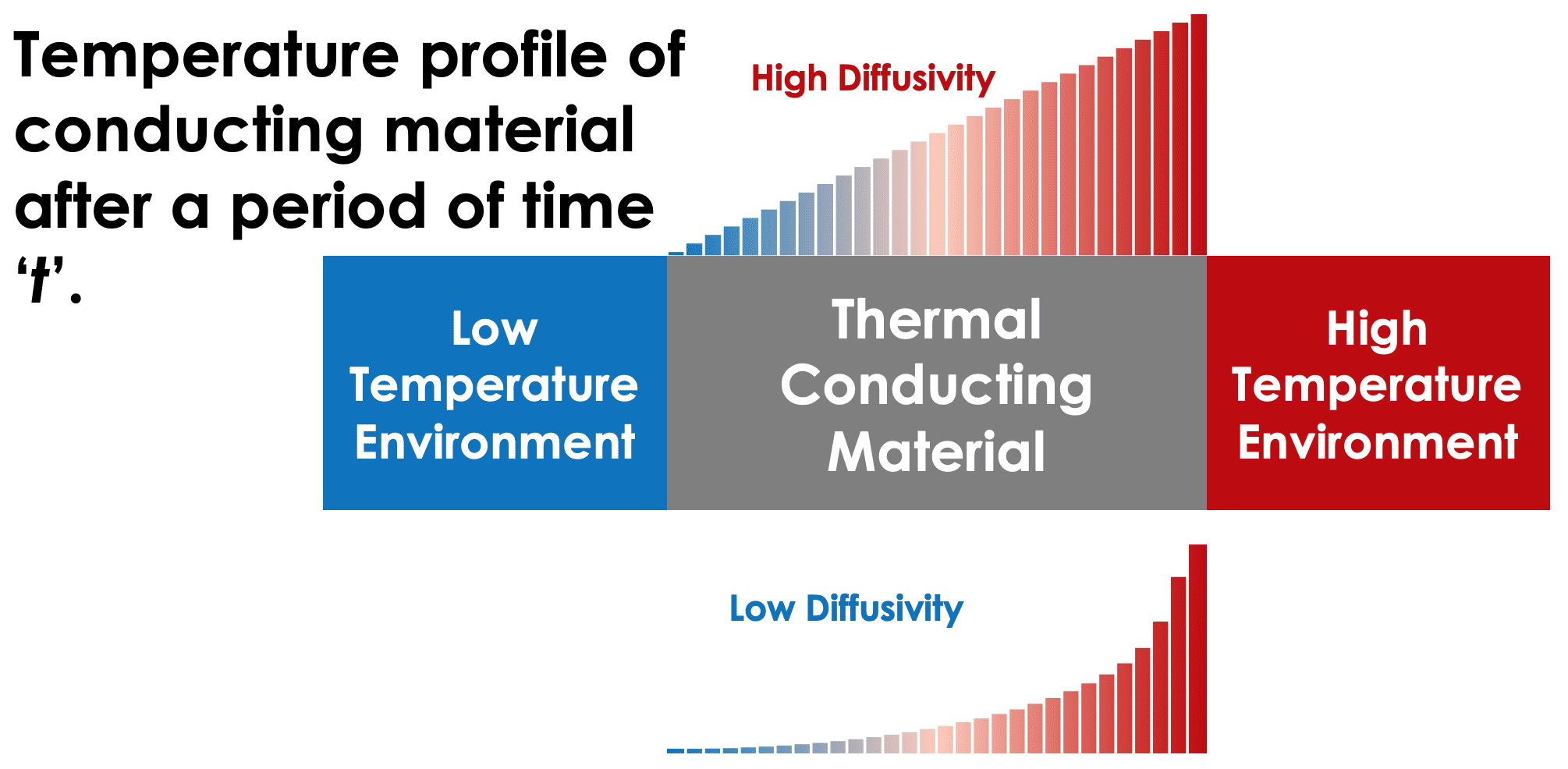
Thermal diffusivity, given by dividing the thermal conductivity of a material by its volumetric heat capacity, can be thought of as the ratio of temperature change throughout the material as it conducts a quantity of heat or the rate at which a thermal equilibrium is reached between the hot and cold end of the material.
A high thermal diffusivity indicates the material will rapidly equilibrate, whereas a low diffusivity indicates it is more likely to increase its internal temperature as heat flows through. The SI units of thermal diffusivity are metres squared per second m2/s. Insulation material for both buildings and textiles help prevent the loss of heat from a room, object or person and will typically have a low diffusivity.
Thermal Effusivity
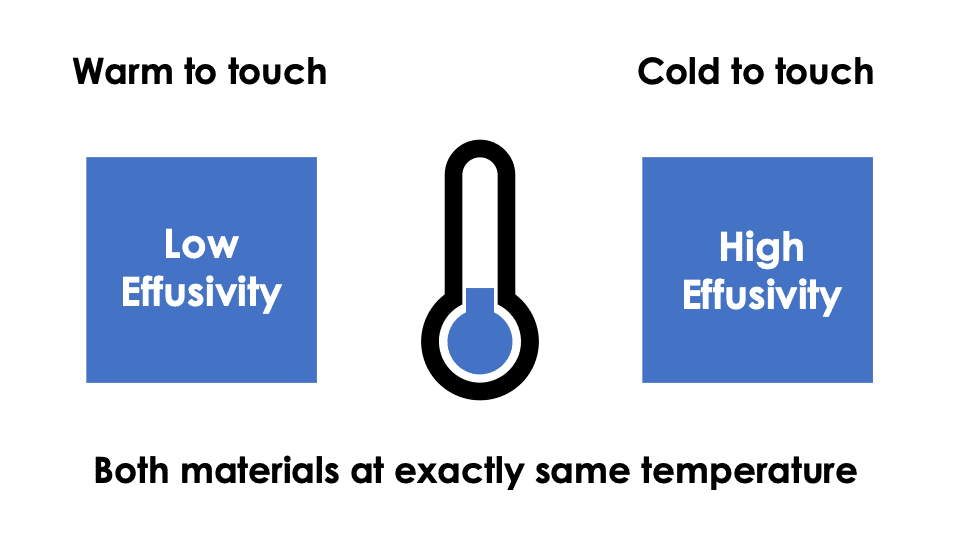
Thermal effusivity, in simple terms, is a measure of the material’s ability to exchange thermal energy with its surroundings at a surface. A high thermal effusivity indicates a material is effective at exchanging heat with its surroundings, whereas a low thermal effusivity indicates a relatively low capability to exchange thermal energy.
Thermal effusivity is the square root of the product of thermal conductivity, density and specific heat capacity – (kρCp)1/2 – with SI units of J/(m2.K.s1/2)
Thermal effusivity can be used to benchmark the heat management of textiles – for example, in sportswear the textile used must be able to keep the wearer warm in a cold environment, but allow the wearer to cool off when sweating. This is often facilitated through a clever combination of thread density, designing pores into the weave of the fabric and choosing specialty materials with targeted thermal properties.
Heat Capacity
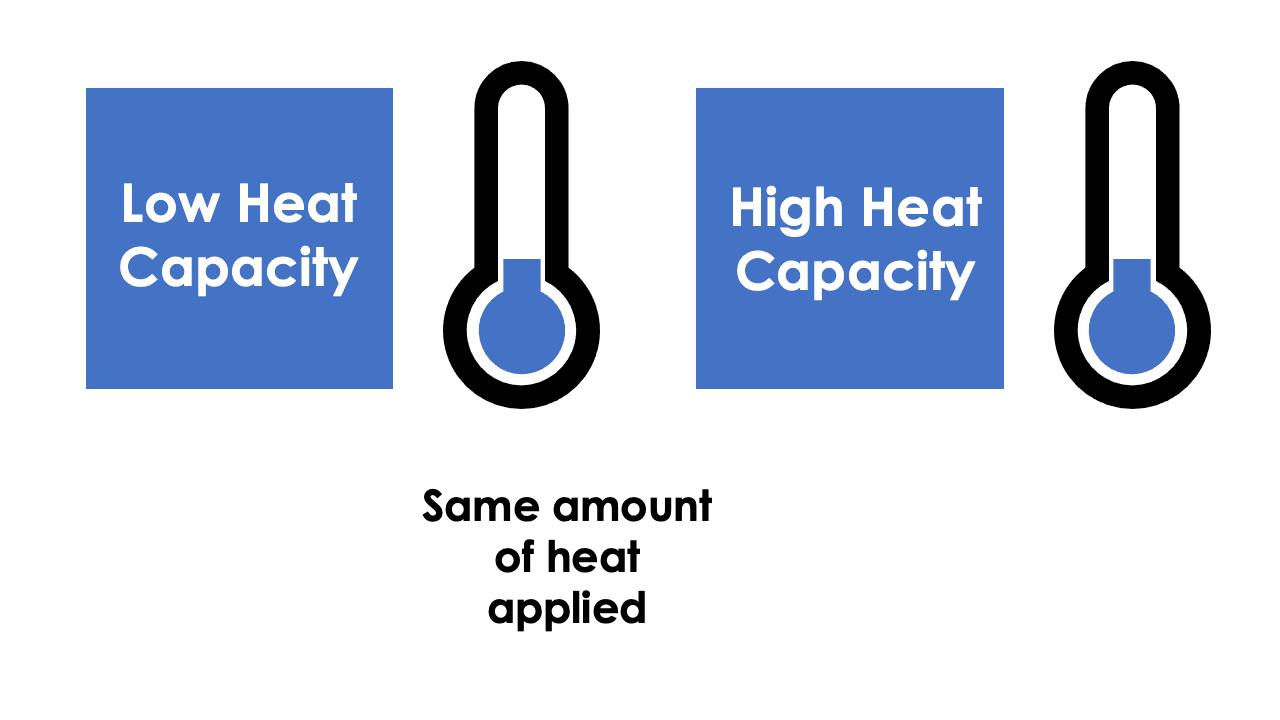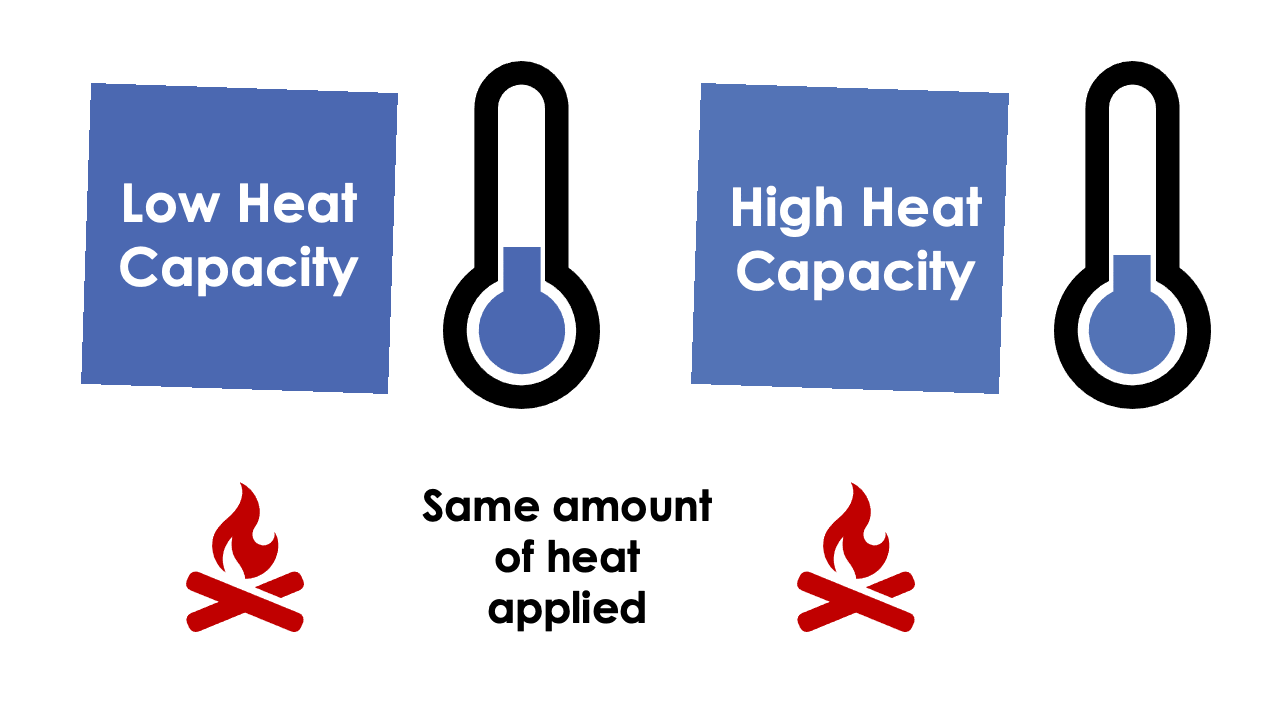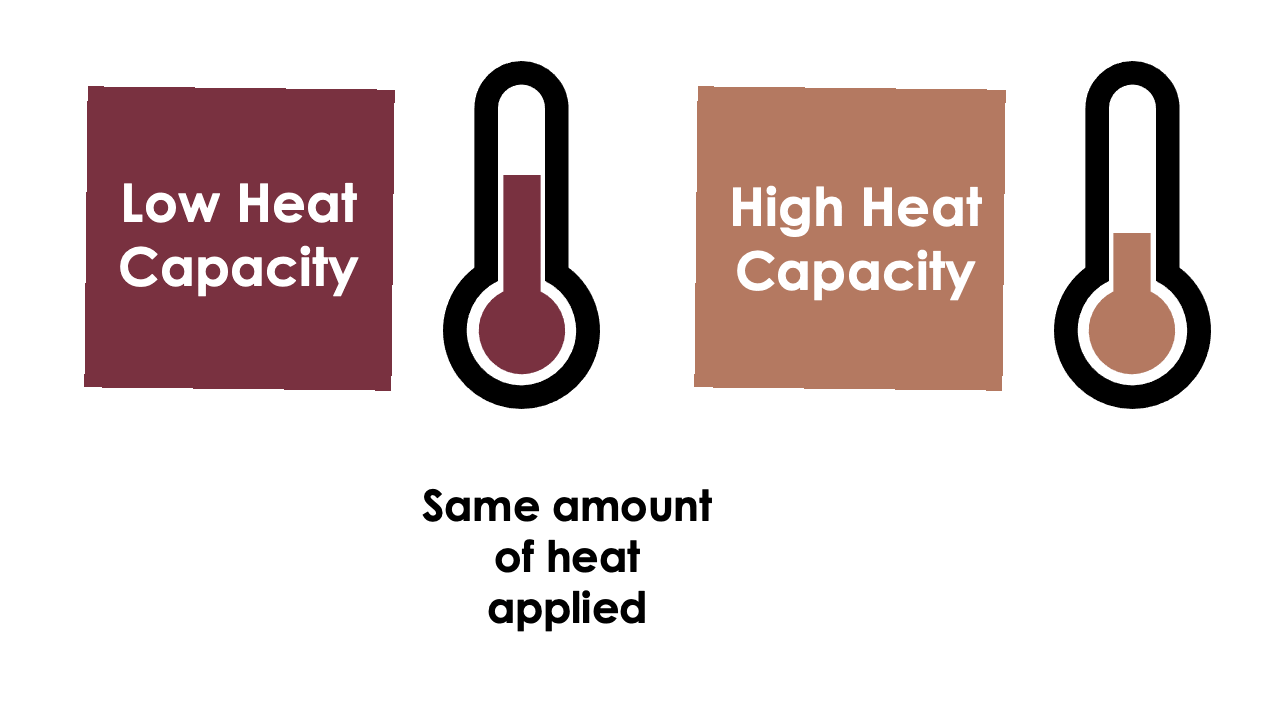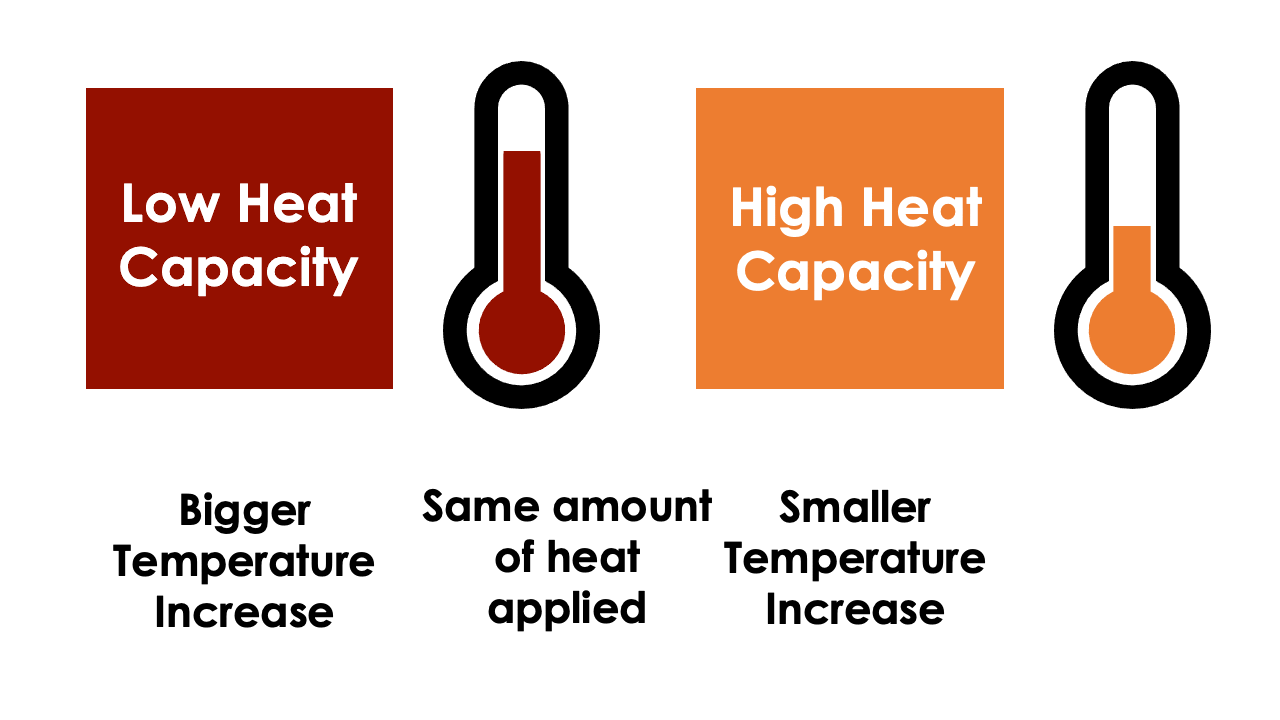
The temperature of a material is the manifestation of the amount of thermal energy it possesses and is heavily influenced by the material’s density and heat capacity. The heat capacity of a material is often given with reference to either density (specific heat capacity) or volume (volumetric heat capacity). Specific heat capacity can be thought of as the effort or work required to increase the temperature of one unit of mass of material by one unit of temperature, the SI unit being Joule per kilogram per Kelvin. When you multiply the specific heat capacity by the density of the material, you can calculate the volumetric heat capacity, which is the effort or work required to increase the temperature of one unit volume of material by one degrees Kelvin, the SI unit being Joule per metres cubed per Kelvin.
If a material has a low specific heat capacity, then a relatively low amount of heat is required to increase the temperature of the material conversely, a high specific heat capacity material will require comparatively more energy to raise the temperature by the same amount. This can be useful within the building materials industry when trying to design buildings capable of protecting its inhabitants from environmental temperature changes.
Whether you are looking to better understand the efficiency of heat transfer in your material, for managing heat with thermal interface materials, to ensuring unit processes are as efficient as possible or simply ensuring that your material will behave and perform as predicted, our lab has a number of complimentary capabilities for characterising the physical properties of soft materials.
Let us know if you had any questions about thermal conductivity, specific heat capacity or other complimentary physical characterisations.
